Government-Funded Randomized Controlled Trial Published in Annals of Internal Medicine Showcases Evidence Generation Through Strategic Clinical Collaboration
Ursodeoxycholic acid (UDCA), the active ingredient in Daewoong Pharmaceutical’s liver function improvement drug Ursa, showed promising symptom improvement signals in early-stage COVID-19 long-haul patients, according to results from a clinical trial operationally supported by BXPLANT. Notably, treatment efficacy varied according to time elapsed since infection, suggesting that “timing of intervention” may be a critical variable in treating post-COVID conditions.
Daewoong Pharmaceutical announced on the 9th that UDCA demonstrated symptom improvement signals in patients within 2-6 months post-COVID-19 infection. The research results were published in the international medical journal Annals of Internal Medicine.
Early Intervention Shows Significant Benefits
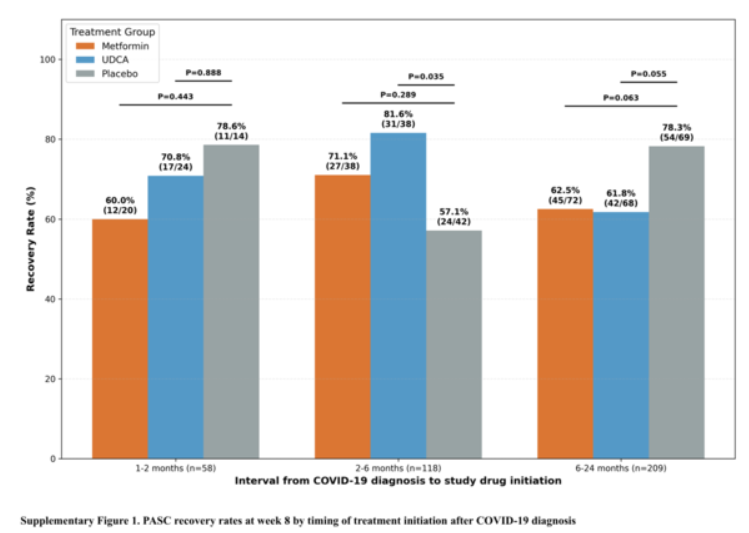
Study results revealed that among patients 2-6 months post-infection, the symptom improvement rate in the UDCA treatment group reached 81.6%, significantly higher than the placebo group (57.1%)—approximately 43% greater improvement on a simple comparison basis. However, in patients more than 6 months post-infection, no significant difference was observed between UDCA and placebo groups.
Researchers explained that these results suggest the timing of therapeutic intervention may be an important variable in treating COVID-19 long-haul syndrome. The findings represent an exploratory confirmation of pharmacological treatment potential in patients at a relatively early stage post-infection.
Inflammation Markers Support Clinical Observations
The research team analyzed inflammatory responses alongside symptom changes. Patients experiencing symptom improvement showed a tendency toward decreased inflammation-related markers, with these changes more prominently observed in the 2-6 month post-infection patient group. However, researchers noted that additional analysis is needed to determine whether these changes resulted from drug effects.
COVID-19 long-haul syndrome, characterized by prolonged fatigue, dyspnea, and cognitive impairment following infection, has been identified as a public health concern by the World Health Organization (WHO) and other major health authorities. Currently, treatment focuses primarily on rehabilitation and symptom management, with limited clinical evidence for pharmacological interventions.
Collaborative Research Model
This study was conducted as a Korea Disease Control and Prevention Agency (KDCPA) research project and represents a domestic randomized controlled trial with operational support provided by BXPLANT. Professor Sung-Han Kim from the Division of Infectious Diseases at Asan Medical Center served as principal investigator, with joint participation from Asan Medical Center and Hallym University Kangnam Sacred Heart Hospital. The study was conducted from July 2024 to March 2025 with COVID-19 long-haul patients, comparatively evaluating the therapeutic potential of UDCA and metformin.
Professor Kim stated, “COVID-19 long-haul syndrome remains an area without established standardized pharmacological treatment strategies. This study demonstrates, through results observed in specific patient time-window cohorts, the necessity for treatment timing-based approaches and additional clinical research.”
BXPLANT’s Role: Strategic Clinical Operations for Evidence Generation
This successful publication exemplifies BXPLANT’s capabilities in executing government-funded clinical research projects and generating high-quality evidence through strategic clinical collaboration. By providing comprehensive operational support—from study design optimization and regulatory compliance to data management and publication-grade documentation—BXPLANT enabled the research team to deliver rigorous scientific evidence addressing an unmet medical need. This case demonstrates our commitment to advancing therapeutic innovations through meticulous clinical trial execution and our ability to bridge academic research excellence with regulatory-grade clinical operations, ultimately contributing to evidence-based treatment strategies for emerging health challenges.
source : ‘Positive Signal for Early Intervention of UDCA for COVID-19 Long-Haul Syndrome”, Ji-Soo Park, Sedaily, 2026.03.09., https://www.sedaily.com/article/20016719

